Tips and tricks to design and test qPCR primers – advanced procedure
Design and test your own qPCR primers
- Find the NCBI Gene page for the gene you would like to study. For example, for Human GAPDH.
- Scroll down to the mRNA and Protein(s) section of the NCBI gene page and determine how many mRNA variants are listed:
- For example for Human GAPDH, there are two mRNA variants: NM_001256799 and NM_002046.
- Decide which mRNA variant(s) you would like to study. If you would like to study only one variant, go to step 4a, otherwise skip to step 4b.
- Use Primer-Blast to design primers specific for a single mRNA variant:
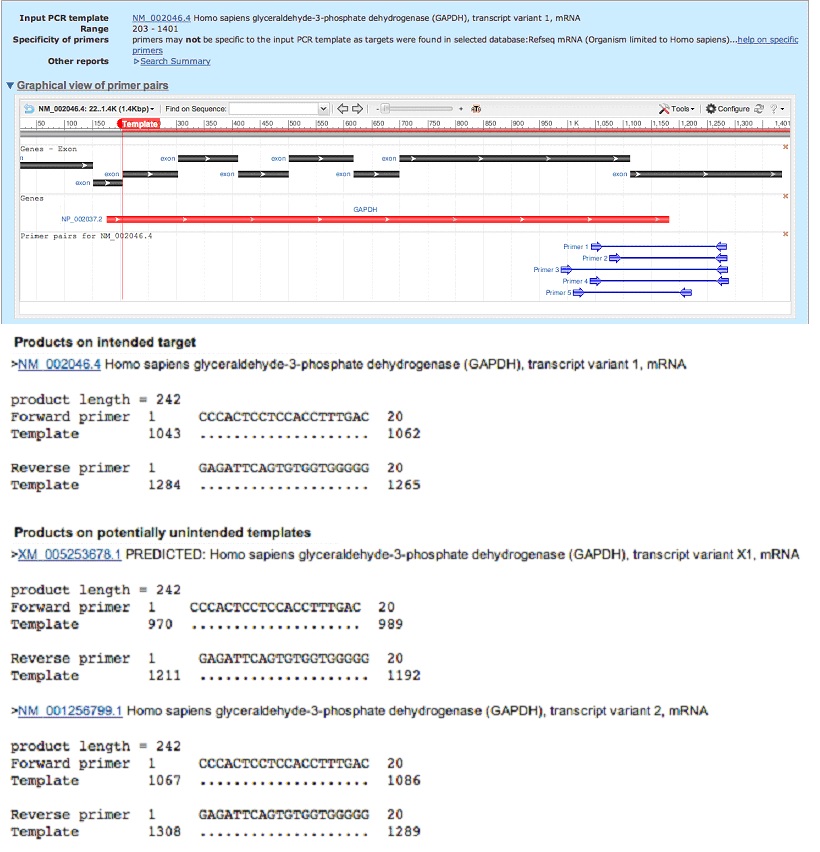
- In the box “Enter accession, gi, or FASTA sequence” paste the mRNA variant accession (for example: NM_002046).
- For PCR Product Size, set the Min=90 and the Max=300.
- Check the Intron Inclusion box: Primer pair must be separated by at least one intron on the corresponding genomic DNA.
- Under Primer Pair Specificity Checking Parameter change the organism to whatever organism you are studing (e.g. Homo sapiens, Mus musculus).
- Check the box “Show results in a new window“.
- Click “Get Primers“.
- Confirm that the primers do not have any “Products on potentially unintended templates” in the output. (Now skip to step 5).
Note: Primer-Blast may fail to find primers that span an intron, so you may have to uncheck this box and search again, however, the resulting primers will also amplify genomic DNA, so DNAse digestion of mRNA should be performed prior to the reverse transcription step of qPCR using a good RNAse-free DNAse).
Example of output aiming at designing PCR primers

5. Use Primer-Blast to design primers to amplify regions from multiple mRNA variants of the same gene:
- Use a clustal alignment site.
- Change the tab from “Protein” to “DNA”.
- Copy-Paste multiple FASTA sequences for the mRNAs you would like to study into the box:
- For example, open the two mRNA variant links NM_001256799 and NM_002046.
- At the top of the pages, click on the FASTA links.
- Copy the FASTA sequences and paste them into the Clustal box one after the other.
- Repeat for more mRNA variant sequences if desired, pasting the new sequence on the line following the previous one.
- Click “Execute Multiple Sequence Alignment“.
Example of output for sequence alignment (beginning and end of the sequences are only shown here)


Note: Here, you can see that the two GAPDH mRNA variants have precisely the same sequences from position 203 to position 1401 of NM_002046.
- In the box “Enter accession, gi, or FASTA sequence” paste the mRNA variant accession (for example: NM_002046).
- Just to the right of that box, enter a range for the forward primer, From: 203, To: 1101.
Note: These values are based on the clustal alignment above-described
- For PCR Product Size, set the Min=90 and the Max=300.
- Check the Intron Inclusion box: Primer pair must be separated by at least one intron on the corresponding genomic DNA;
- Under Primer Pair Specificity Checking Parameter change the organism to whatever organism you are studing (e.g. Homo sapiens, Mus musculus);
- Check the box “Show results in a new window“;
- Click “Get Primers“.
Example of output aiming at designing PCR primers
Note: Here you can see that the proposed primers should amplify regions from multiple mRNA variants, but all will give a product of the same length and sequence.
- Order the proposed Forward and Reverse primers (at least 2 sets) from a provider of pre-designed and pre-tested Primer Sets or of custom PCR primer synthesis.
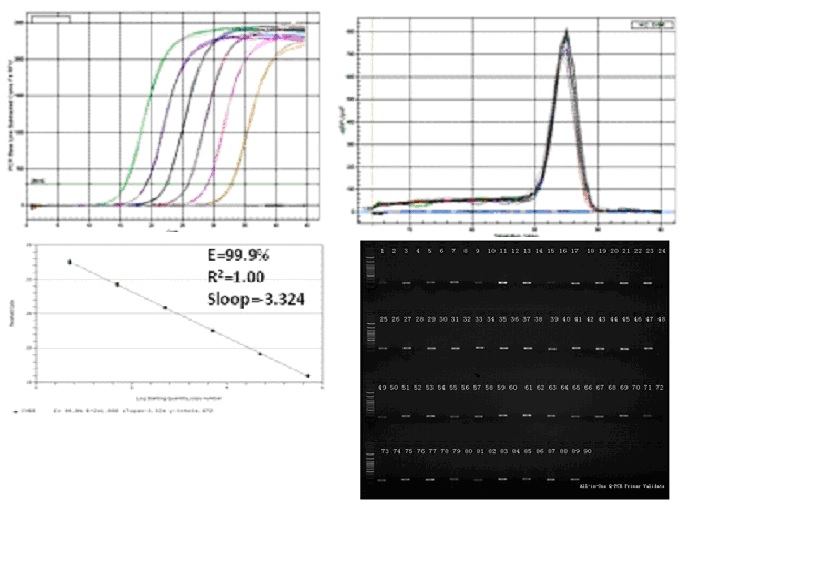
Sample validation data for commercially-validated primers (amplification and melting curves, efficiency plots and EtBr-stained agarose gels demonstrating specificity). Here are examples of qPCR Primers for Human GAPDH (HQP006940) and murine GAPDH (MQP027158).
- Primers will generally arrive lyophilized (dry) with instructions for how much water or TE buffer to add for 100uM solution. Dilute the forward and reverse primers separately to 100uM with 1X TE Buffer, pH8.0.
- Mix together 800uL Nuclease-Free Water 100uL forward primer and 100ul reverse primer for a final working stock of 10uM forward and reverse mixture. You will use this mixture at a final concentration of 0.4uM for qPCR.



