
Very recently we launched new Live Cell Imaging tools: SiR-Actin and SiR-Tubulin, produced by Spirochrome.
These stains allow you to stain actin and tubulin in living cells without the need to transfect cells – as I described in my previous posts on these tools:
- 2 new Actin and Tubulin live-cell imaging stains – without transfection!
- Verapamil can enhance live cell staining of Actin & Tubulin with SiR-dyes
Today, I invite you to take a look at the brilliant results users of the stains have obtained. Some of them have already been published during the past months.
Most recent publications:
One of the most recent publications using SiR-Actin comes from the lab of the Nobel price winner Stefan W. Hell, who is one of the directors of the Max Planck Institute for Biophysical Chemistry in Göttingen, Germany:
By using STED (stimulated emission depletion) microscopy, Hell’s group could visualize the periodic subcortical actin structure in axons and dendrites in cultured hippocampal neurons. These results were obtained by staining Actin with SiR-Actin.
Another recent paper even brought an image of HEK293 cells co-expressing CaVβ and CaV1.2 L-type calcium channel and stained for actin filaments using SiR-Actin straight to the front cover of the respective JBC issue.
The group around Patricia Hidalgo at the Institute of Complex Systems in Juelich, Germany, could show that the β-subunit (CaVβ) n of cardiac L-type calcium channels associates directly with actin filaments – again SiR-Actin was successfully used to get these results.
Videos showing the use of SiR-Tubulin
As SiR-stains are very photo stable and do not show toxic effects, even in long-term incubation, they are excellent tools for visualizing biological processes over time in videos.
We show here an example of dividing HeLa cell expressing mcherry-H2B (red) stained with SiR-Tubulin (green). Data have been collected by confocal imaging (Courtesy of Daniel Gerlich and Claudia Blaukopf, Institute of Molecular Biotechnology, Vienna, Austria).
Another video shows newborn mouse primary cardiac myocytes stained with SiR-actin. The authors used high speed (50fps) confocal imaging (Courtesy of Adam Kwiatkowski and Simon Watkins, Department of cell biology and center for biologic imaging, University of Pittsburgh, US).
Results provided by our customers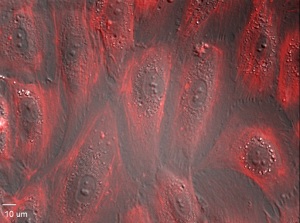
HUVEC monolayer, stained with SiR-Actin. A ZEISS Axiovert 200 Marianas inverted microscope with custom ZEISS 40x air lens was used (Courtesy of Erik T. Valent and Geerten P. van Nieuw Amerongen, VU Medisch Centrum, Amsterdam , The Netherlands).
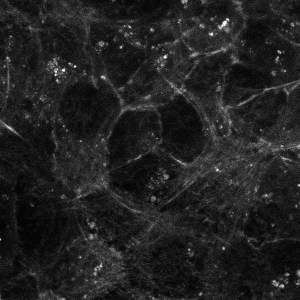 Human endothelial cells B4G12 were grown on Ibidi® µ-Slide 4 Well dishes at confluence. Cells were labeled for 2h at 37°C/5% CO2 with 0.2 mM SiR-Actin and a confocal image was acquired exciting with a Laser Helio Neon of 637 nm with a Zeiss Confocal LSM510 META system (Courtesy of Cristina Ortega Muñoz and Jaime Millan, Centro de Biologica Molecular, Madrid, Spain).
Human endothelial cells B4G12 were grown on Ibidi® µ-Slide 4 Well dishes at confluence. Cells were labeled for 2h at 37°C/5% CO2 with 0.2 mM SiR-Actin and a confocal image was acquired exciting with a Laser Helio Neon of 637 nm with a Zeiss Confocal LSM510 META system (Courtesy of Cristina Ortega Muñoz and Jaime Millan, Centro de Biologica Molecular, Madrid, Spain).
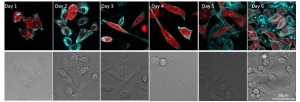
SiR-Actin staining for transient labeling of breast cancer cells implanted into a xenogeneic zebrafish host. SiR-Actin stained cells (MDA-mb231B1 dsRED), stained overnight, followed for 6 days via confocal imaging (Leica TCS SPE) microscope (63x objective). Note the retention of SiR-Actin in vitro, and the absence of cellular artefacts with concentrations of SiR-Actin below 100 nM. Cytosolic CMV driven dsRED shown in red and Sir-Actin shown in cyan (Courtesy of Arwin Groenewoud and B. Ewa Snaar-Jagalska, Institute of Biology, Leiden, The Netherlands).
 Primary neurons derived from cortex + hippocampus of wild type mouse C57BL/6J were cultured for 19 days and subsequently stained with SiR-Actin and visualized with a Nikon NSTORM to shown spine formation (Courtesy of Oxana Klementieva, Gunnar Gouras (Lund University) and Catherine Kitts from Lund University Bioimaging Center [LBIC], Sweden).
Primary neurons derived from cortex + hippocampus of wild type mouse C57BL/6J were cultured for 19 days and subsequently stained with SiR-Actin and visualized with a Nikon NSTORM to shown spine formation (Courtesy of Oxana Klementieva, Gunnar Gouras (Lund University) and Catherine Kitts from Lund University Bioimaging Center [LBIC], Sweden).
We would like to thank all the users who provided the pictures and results!
And of course, we invite all researchers who would like to test the SiR stains in their laboratory to contact us through the form sheet below.
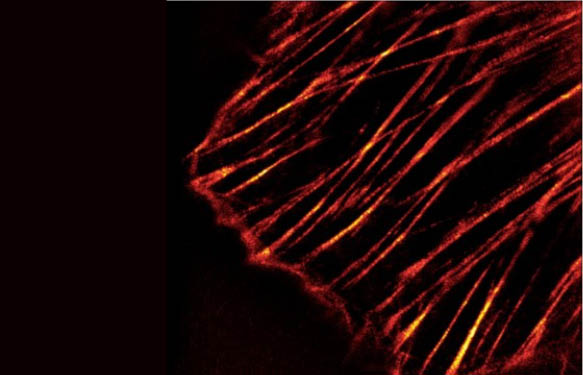



17 Responses
Our lab is interested in trying siR-actin. Is there a source to get some free sample to try on?
Hello Ting,
I will contact you through email with a concrete quote.
Best regards
Ali
I’m interested in testing the siR-actin dye in a live cell imaging experiment. Is there a way to get a sample for testing in stem cells?
Dear Francesca,
we do not have samples sizes of the SiR.stains available, but we will send you a quote for the SiR-actin stain.
Best regards
Ali
Hello there,
I am too, interested in this dye, to label neurons/astrocyte and follow their cytoskeletal dynamics.
Could you please contact me for a quote?
I must first try out to assess the retention time, cell density etc.
Many thanks, look forward to hearing from you
Sam
Dear Sam,
You are going to be contacted by a colleague regarding your request.
Regards,
Philippe
Hi there,
I’m interested in using this to visualise cytoskeletal dynamics in primary neutrophils. Could you let me know the feasibility of this and provide a quote for a sample?
Thanks,
Jessie
Dear Jessie,
Thank you for reading our blog.
These dyes have been used on various types of blood cells. Please let us investigate if we have information regarding neutrophils in particular. You will be contacted by a member of our team very soon.
Regards,
Philippe
Hi,
I am looking for siR-Tubulin GFP live cell dye. Do you make it?
Good afternoon,
First of all, thank you for your interest on this post.
In fact SiR Tubulin is a fluorescent dye by itself (λabs of 652 nm and a λem of 674 nm in the far red), so it can be directly used for live cell imaging of tubulin in cells.
If you want to label Tubulin with GFP and so one a green color, we can discuss more in detail about your experiement to see if we can find a solution.
Don’t hesitate to contact me directly to discuss about the SiR Tubulin probe.
Regards,
Frederic
phone:+33130463986
mobile:+33673976846
mail: frederic.samazan@tebu-bio.com
SiR probes produtcs: http://www.tebu-bio.com/brandsearch/265/Spirochrome_AG.html
Hi,
I was wondering if anyone has used the SiR-Actin dye on zebrafish? Also, it would be great if you can contact me and give a quote.
Best,
Agnik
Dear Dr Dasgupta,
First of all I would like to thank you for your interest in our blog and also in the SiR- probes.
I can confirm you that SiR Probes are working on Zebrafish (https://spirochrome.com/faq/ – 7 Tissues and organisms).
Unfortunately tebu-bio company is a unique distributor of Spirochrome products but only in Europe. I will forward your request to the US distributor of Spirochrome products who will contact you to provide you a quote.
Best,
Frédéric
Hello,
I am interested in the applications of SiR-Tubulin to replace the easily bleached Taxol dyes in live cell imaging experiments. I was wondering about it’s feasibility in terms of labelling alpha tubulin in primary cilia of osteocytes. Also if you could advise me on the cytotoxicity of this dye and the applicability with the Calcein AM stain (green).
Cheers,
Bilawal
Dear Bilawal,
First of all, I would to thank you for your interest in our post and products.
As you can see in this publication (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5858867 ) the Sir Tublin can be used to label Microtubule in Osteocyte. Used under the concentration mentionned in the data sheet, this Sir Probe don’t have any cytotoxic effect on the microtubule polymerisation and of course can be used with teh Calcein AM stain, as this probes presents different wavelengths. THe only important point for the SIR -Tubulin probe, is that this probe can only be used fo rlive imaging and not on fixed cells.
As we are the unique distributor of spirochrome product only in Europe, I sent you mail adress to the distributor of this SiR probes for Canada.
Have a good day,
Best regards,
Frederic
HI I am interested in using the SiR-tubulin for looking at microtubule dynamics in cortical neutrons isolated from our disease model. Could you please get in contact about a quote/sample of the dye?
Thanks,
Allie
Dear Dr Colaco,
First of all, I would like to thank you for reading our post. Secondly, I would like to inform you that I have sent your mail address to our local tebu-bio office (https://www.tebu-bio.com/cms/1062/.html) who will get back to you with a proposal for this product.
Thank you and have a good afternoon,
Best regards,
Frédéric